Rechargeable batteries have been widely applied in all aspects of our life. Most rapidly developed and widely used lithium-ion batteries (LIBs) cannot meet the large-scale energy storage systems because lithium resources are expensive and geographically constrained.
Sodium-ion batteries (NIBs) have attracted increasing attention because of the abundant resources of sodium. The polyanion-type sodium vanadium fluorophosphates Na3(VO1-xPO4)2F1+2x (0≤x≤1) (NVPFs), possess an energy density of around 480 Wh/kg. If NVPFs could be industrially used in NIBs, the energy density is comparable to LIBs.
A research group led by Dr. ZHAO Junmei from the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences proposed a concept of integration of extraction-separation and material-preparation. They developed a facile one-step room-temperature strategy after putting forward a series of hydro-/solve-thermal synthetic routes for the synthesis of Na3(VOPO4)2F (NVPOF).
NVPFs now are synthesized using a solid-state method, and the manufacturing operations under high temperature occupy a large part of their cost. Therefore, developing low-temperature solution routes for NVPFs is the key for industrial application.
This strategy can significantly decrease the manufacturing cost of electrode materials. The obtained NVPOF microspheres have a three-dimensional multi-shelled hierarchical architecture, based on the in situ generated bubbles as soft-templates and the self-assembly of nanoparticles on bubbles.
This is the first time to report a rapid and facile large-scale room-temperature controllable synthesis of a polyanionic compound. The synthesized NVPOF, without any additional treatments, delivers superior rate capability and outstanding cycling stability.
While the soft-template strategy could be extended to synthesize other multi-shelled architectures. The in situ generated bubbles as soft-templates are also distinctive compared to the common hard-templates and surfactant-templates.
This indicates that the technology can greatly decline the manufacturing cost of cathode materials. The strategy could be used for recycling waste rechargeable batteries and be extended to synthesize other similar electrode materials.
"The novel room-temperature synthesis for polyanionic compounds is a milestone technology that subverts the conventional ceramic and hydrothermal methods." Dr. Zhao said, "Simultaneously, the research results are bound to accelerate NVPFs' industrialization process."
The study entitled "Scalable room-temperature synthesis of multi-shelled Na3(VOPO4)2F microsphere cathodes" was published in Joule (doi: 10.1016/j.joule.2018.07.027).
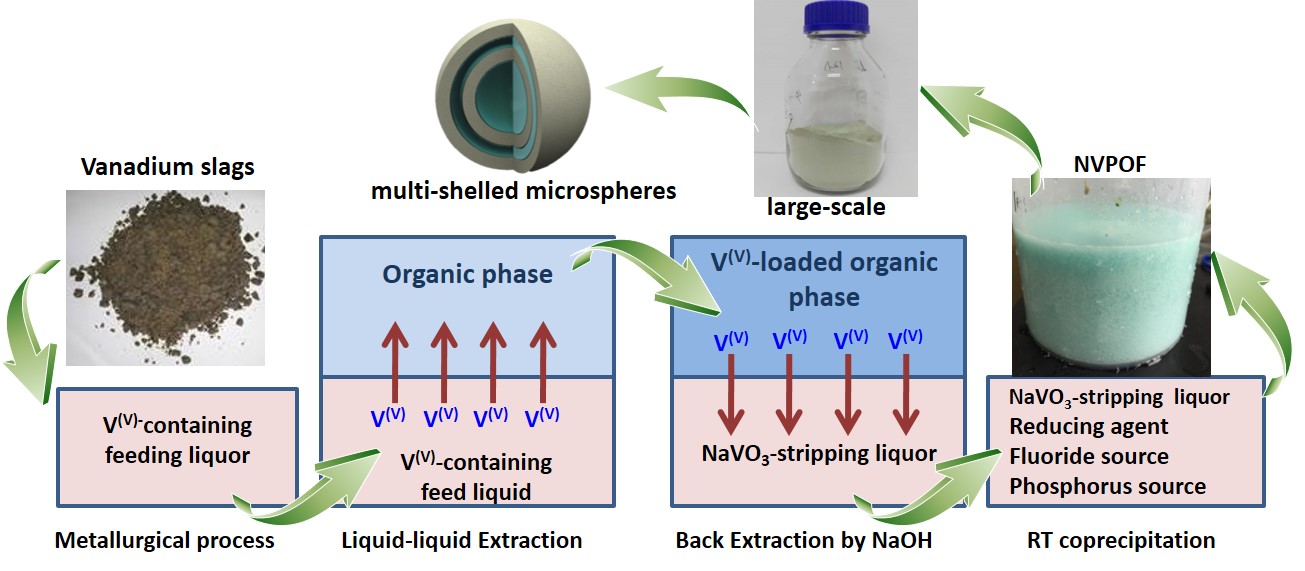
Fig 1 Schematic of integration of extraction-separation and material-preparation (NVPOF as an example)(Image by ZHAO Junmei)
Media Contact:
LI Xiangyu
International Cooperation Office, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, P. R. China.
E-mail: xiangyuli@ipe.ac.cn
Tel: 86-10-62551358