The development of an artificial photosynthesis system that converts CO2 into a kind of chemical energy with high efficiency is one of the major issues to solve energy and environmental problems. Electromicrobial conversion of CO2 is a promising alternative to the direct transfer of CO2 into biomass by using bacteria. The bacteria consume H2 and CO2 for living. The efficiency of such a hybrid system strongly depends on the electrocatalyst used for water-splitting to produce H2. To achieve high conversion efficiency, the electrocatalyst must be stable, with low overpotential and high biocompatibility during the electrolysis. A research team led by Prof. Zhang Guangjin from the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences evidenced that a mixed noble metal free Co/Cu-containing polyoxometalate/Carbon Cloth (Cu6Co7/CC) hybrid possesses excellent activity as a water splitting electrocatalyst at neutral pH. The long term stability of the activated noble metal-free electrodes together with their good biocompatibility -- a negligible amount of hydrogen peroxide being produced during the electrolysis process -- made it suitable for applications in biosynthetic systems. CO2 conversion with bacteria and the Cu6Co7 catalyst as both anode and cathode materials has been tested. The hydrogen produced at the cathode was the sole source of biological reducing agents. The growth of the bacteria and the accumulation of biomass were monitored by UV-vis spectroscopy. It can be obtained by calculation that the overall solar-to-biomass efficiency of this hydride system can reach 10%, which is nearly 10 times higher than the performance of natural plants. Their findings entitled “An overall water-splitting polyoxometalate catalyst for the electromicrobial conversion of CO2in neutral water” were published in Journal of Materials Chemistry A. This work was supported by the National Natural Science Foundation of China and Youth Innovation Promotion Association of CAS. 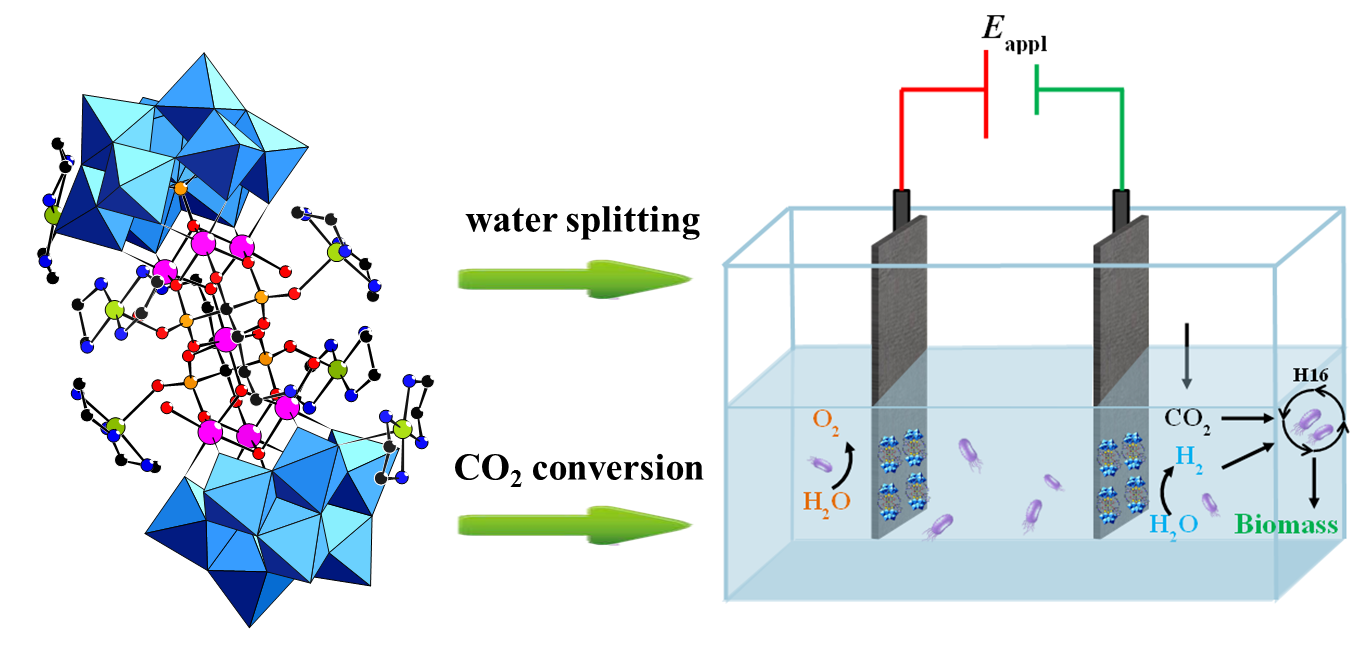
Figure 1 Schematic illustration of the bioelectrochemical system for CO2 fixation. (Image by ZHANG Guangjin et al.) Media Contact:
LI Xiangyu
International Cooperation Office, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, P. R. China.
E-mail: xiangyuli@ipe.ac.cn
Tel: 86-10-62551358
|