Alkaline rechargeable battery, as a sort of advanced energy storage device with higher power density, energy density, superior rate capacity, fast and reversible surface redox reactions, has been widely used in various electric systems. A great amount of researches were focused on transition metal oxide nanomaterials which not only own larger surface area, but a large amount of faradaic capacitance.
However, because of van der Waals force or electrostatic attraction, nanoparticle is easy to self-aggregate which decreases the contacting area between electrolyte and active material named electrochemically active surface area (EASA) and increases the resistance of bulk solid-state diffusion. And the pore is a very important factor limiting the diffusion of electrolyte from outside to inside of materials which is requirement for redox reaction. Therefore, controlling the architecture of electrode material to increase EASA plays an important role in enhancing rate capacity. Fortunately, multi-shelled hollow sphere material can conquer above disadvantages because of unique structure.
Recently, researchers further developed the sequential templating method to prepare multi-shelled hollow sphere by adjusting the crystalline temperature of the product. As shown in Fig 1, sextuple and septuple-shelled hollow microspheres with thin shell, uniform gap between adjacent shells and large specific area are synthesized by finely controlling the molar ratio of Co/Mn to realize the control of crystalline rate, which is quite distinctive. Due to the synergistic effects of (Co2/3Mn1/3)(Co5/6Mn1/6)2O4 complex oxide and its unique structure properties, including high specific area, large pore volume, which provide the hollow spheres with more active sites and facilitate transport of the electrolyte, excellent charge storage performance and prominent mechanical stability are obtained. When used as electrode materials of alkaline rechargeable battery (Fig 2), compared with others, septuple-shelled hollow microspheres demonstrated an excellent specific capacity of 236.39 mAh g-1 at a current density of 1 A g-1 by three-electrode system (106.85 mAh g-1 at 0.5 A g-1 in alkaline rechargeable battery) and excellent cycling life (96.07% capacity retention after cycling 2000 time at 8 A g-1 and 84.17% capacity retention after cycling 3000 time at 1 A g-1 for alkaline rechargeable battery). This new strategy is expected to be broadly employed to synthesize other multi-shelled hollow complex oxides, which have promising applications in future energy and environmental fields.
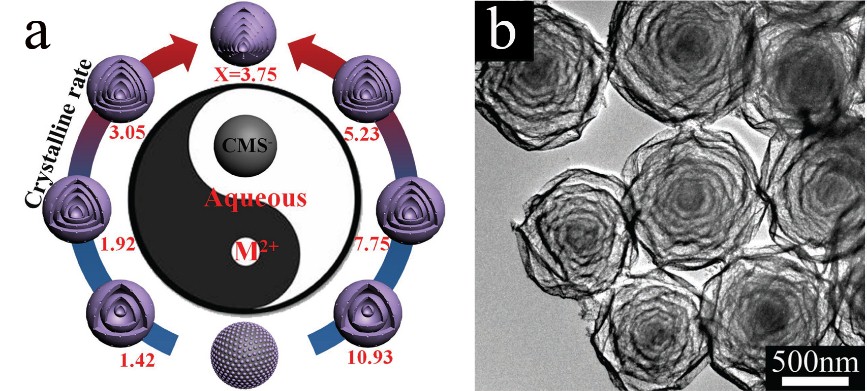
Fig 1. a) Septuple-shelled hollow microspheres are synthesized by finely controlling the molar ratio of Co/Mn using sequential templating method (M2+: Co2+ and Mn2+; X: the molar ratio of Co/Mn); b) TEM of septuple-shelled (Co2/3Mn1/3)(Co5/6Mn1/6)2O4 hollow spheres.
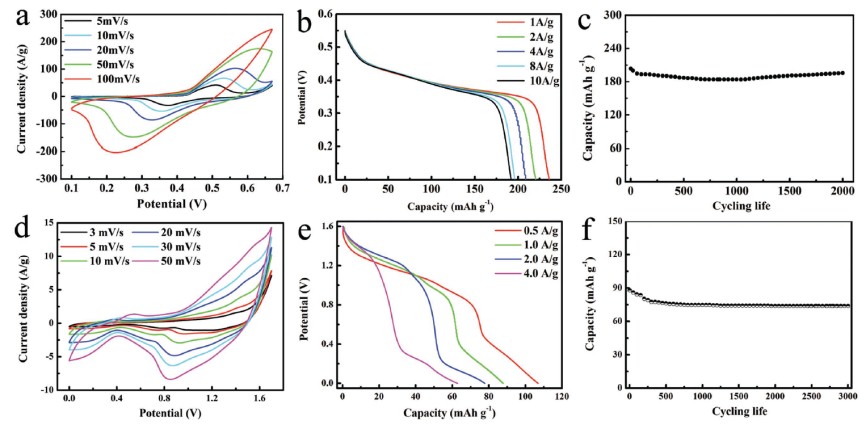
Fig 2. The electrical performance of septuple-shelled (Co2/3Mn1/3)(Co5/6Mn1/6)2O4 hollow sphere as electrode material in three-electrode system: a) cyclic voltammetric; b) galvanostatic charge-discharge voltage; c) cycling life and alkaline rechargeable battery: d) cyclic voltammetry curves; e) galvanostatic charge-discharge curves; f) cycling stability.

Fig 3. Inside back cover of Adv. Mater. 2017 Vol. 29 No. 34
The research team includes a group led by Prof. WANG Dan from the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences (CAS), a group led by YU Ranbo from University of Science & Technology Beijing and a group led by ZHANG Yu from Beihang University. The research was supported by the National Science Fund for Distinguished Young Scholars (No. 21325105), National Natural Science Foundation of China (Nos. 21590795, 21671016, 51472025, 51472244), The National Key Research and Development Program of China (2016YFB0600903), CAS Interdisciplinary Innovation Team, the Foundation for State Key Laboratory of Biochemical Engineering, and et al.
Their work entitled “Formation of septuple-shelled (Co2/3Mn1/3)(Co5/6Mn1/6)2O4 hollow spheres as electrode material for alkaline rechargeable battery” has been published as the inside back cover (Fig 3) in Adv. Mater (2017, 29, 1700550).
http://onlinelibrary.wiley.com/doi/10.1002/adma.201770247/full
Author Contact:
Prof. WANG Dan
State Key Laboratory of Biochemical Engineering, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, P. R. China.
E-mail: danwang@ipe.ac.cn
Media Contact:
Ms. YUAN Pei
International Cooperation Office, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, P. R. China.
E-mail: pyuan@ipe.ac.cn