Due to the rapid aggravation of the global warming and the decreasing availability of fossil fuels, the development of environmentally friendly, sustainable and renewable energies and sources has become an ever-increasing and urgent issue. Supercapacitors are intermediate devices between conventional batteries and dielectric capacitors that can be used in various electric systems. The pseudo-capacitors have drawn great attentions because of their ability to embrace the advantages of both lithium ion batteries (LIBs) (high energy density) and capacitors (high power density). Among various supercapacitor electrode materials, the manganese oxide is one of the most promising candidates due to its high specific capacitance, environmental compatibility and cost effectiveness. Nevertheless, they still suffer from some drawbacks, such as low specific surface area and poor conductivity. Hollow micro-/nano-structured materials can overcome the above drawbacks due to their large specific surface area, short transport length and good structural stability.
The research team led by Prof. WANG Dan at Institute of Process Engineering (IPE) of the Chinese Academy of Sciences had developed a general strategy to prepare multi-shelled metal oxide hollow microspheres by using sacrificial carbon microspheres (CMSs) as the hard templates. By varying synthetic parameters, they had previously demonstrated controllable synthesis of hierarchical multi-shelled hollow microspheres with adjustable nanoparticle subunits and shell structures. (Angew. Chem. Int. Ed. 2011, 50, 2738-2741; Adv. Mater. 2012, 24, 1046-1049; Angew. Chem. Int. Ed. 2013, 52, 6417-6420; Nano Lett. 2014, 14, 6679-6684)
Recently, they further modify their method by precisely controlling the pH value of the precursor solution, thus controlling the negative charge of the CMSs and the positive charge of Mn cations. As a result, multi-shelled Mn2O3 hollow microspheres with controlled shell thickness and shell number, as well as Mn2O3 nanoparticles were achieved. The unique architecture of these designed nanostructures allows efficient use of pseudo-capacitive Mn2O3 nanomaterials for charge storage due to the facilitated transport of both electrolyte ions and electrons, thus rendering them with a new record high supercapacitor performance. Wherein, the as-synthesized triple-shelled Mn2O3 hollow microspheres with thin porous shells show an extremely high specific capacitance up to 1651 F/g at a current density of 0.5 A/g, and remarkable cycling stability with 92% retention after 2000 consecutive cycles. Besides, the rate capability is also impressive, showing a specific capacitance as high as 1422 F/g at a high current density of 10 A/g. We believe that such earth-abundant, low-cost, environment-friendly, and high-performance electrode materials achieved through a scalable and controllable process, can offer great promise in large-scale energy storage device applications.
The results have been published on Adv. Sci. 2014, 1: 1400011. doi: 10.1002/advs.201400011.
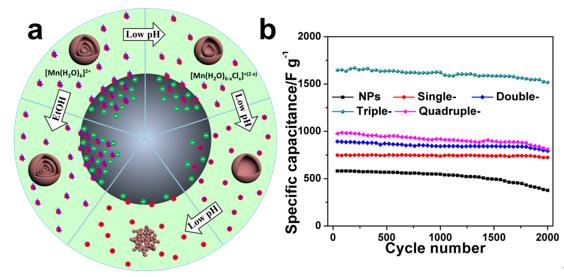
Figure 1 a) Scheme of synthesis mechanism for Mn2O3 nanoparticles and multi-shelled hollow microspheres under different adsorption conditions; b) Cyclic stability of Mn2O3 nanoparticles, single-, double-, triple- and quadruple-shelled hollow microspheres for 2000 cycles at a current density of 1 A/g.
Contact: Prof. WANG Dan at dwang@ipe.ac.cn
Key words: pH-regulated, Supercapacitor, multi-shelled hollow microspheres