Chromium reaches the soil environment through waste disposal emanating from a number of industrial activities, including electroplating, petroleum refining and chromate production and so on, posing a great threat to flora and fauna and persisting for a long time. As a typical chromate production process, the Na2CrO4-NaAlO2-NaVO3-H2O solutions are produced after roasting the chromite with Na2CO3. It is estimated that about each 1000 kg of chromate product (Na2Cr2O7?2H2O) will be accompanied by approximately 200 kg of Cr6+ containing Al(OH)3 residue and 50-100 kg of Ca(VO3)2 residue which contained about 5 kg of highly carcinogenic CaCrO4, resulting in serious environmental pollution and waste of aluminum and vanadium resource. Prof. ZHENG Shili’s research group at Institute of Process Engineering (IPE) of Chinese Academy of Sciences, recently reported a mesoporous ?-AlOOH which was synthesized by reacting NaAlO2 with H2SO4. The synthesized mesoporous ?-AlOOH had a maximum V(V) adsorption capacity and a high adsorption selectivity in the mixture solutions of V(V) and Cr(VI). The mono-oxo, V=O terminal double bonds within VO4 units of vanadate ions were observed to connect oxygen of the coordinatively unsaturated AlVI centres of the mesoporous ?-AlOOH. The mesoporous ?-AlOOH synthesis and in situ V(V) ion adsorption were used in the Na2CrO4–NaAlO2–NaVO3–H2O solution of the chromate production, showing that the V(V) ions were removed effectively without introducing Ca2+ so that the CaCrO4 containing Ca(VO3)2 residue was eliminated. High added value of V2O5 and mesoporous ?-AlOOH could be easily obtained by the V(V) ions containing mesoporous ?-AlOOH. At last, green separation of V(V) and Cr(VI) to reduce resources of waste could be expected. The research article entitled “The vanadate adsorption on a mesoporous boehmite and its cleaner production application of chromate” has been published in Green Chemistry (2014, 16, 4214-4222). The work was supported by the National science Foundation of china (no. 51204254) and chromate production enterprises. 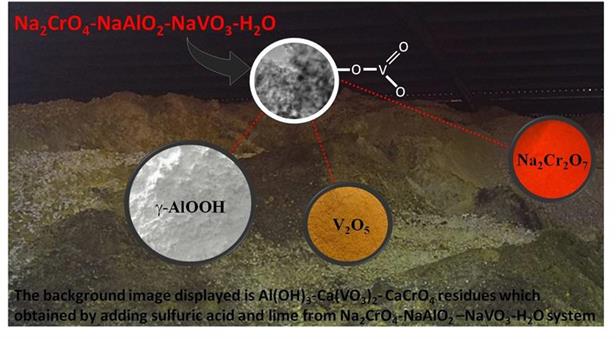 Key words: Boehmite (g-AlOOH), Adsorption, Vanadate, chromate production, Na2CrO4-NaAlO2-NaVO3 aqueous solutions system Contact:: Prof. LI Ping Li, E-mail: lipinggnipil@ipe.ac.cn, Tel:13718635790
|